INTRODUCTION
EEG datasets are destined to be a pillar of precision medicine
Precision medicine is coming in on the back of electroencephalogram (EEG) datasets and the biomarkers they throw up. As R&D units in healthcare and pharmaceutical companies launch EEG dataset projects, private equity funds are backing digital biomarker startups foraying into digital healthcare product development projects.
Originally used as a diagnostic tool for epilepsy, recent advances in artificial intelligence and machine learning have made it possible to explore the use of EEG datasets for:
- Early detection and monitoring of neurological disorders like dementia, Alzheimer’s disease, and Parkinson’s disease.
- Accurate diagnosis and personalized treatment plans for mental health conditions.
- Real-time adjustments to anesthesia dosages to reduce complications.
- Better management of pain in chronic and life-threatening diseases with predictive models.
- Monitoring of sleep patterns, stages, and sleep-related disorders to improve therapies.
- Validation of a drug through pharmacodynamics and pharmacokinetics modeling and simulation during the development stage.
DRIVERS
Technology is giving impetus to the search for novel EEG biomarkers
The rapid development and proliferation of technologies are driving the use of EEG biomarkers to alleviate human suffering and disease.
High-performance computing (HPC)
Artificial intelligence (AI) built on neural networks is capable of analyzing a diverse set of data -images, videos, sensor-generated data, sequential biomarker data, clinical outcomes, and patient demographics. Minus the bias of the human brain, HPC converts computing power into high-performance solutions.
Machine learning (ML)
ML algorithms can scan long hours of EEG data with advanced signal processing and analysis to identify complex patterns and unique biomarkers associated with different medical conditions. Predictive analytics foretell disease progression, assisting physicians in making informed decisions.
Open-source EEG platforms
Large datasets of EEG are accessible for research through online EEG repositories, patient-specific data banks, and toolsets. Computer-based assessments help differentiate between disorders.
Brain-Computer Interface
Assistive devices and applications are available to immobile patients to help them communicate, move and regain independence with the control of a computer cursor through their thought process alone.
Wearables
Compact and wearable devices track sleep patterns, monitor brain waves, detect fatigue in drivers, and apprehend emotions. Conceived in digital healthcare product development projects, these portables are also used for entertainment, gaming, fitness, and learning.
Wireless connectivity
Sensor-based remote monitoring has got a fillip with the advent of 5G, MIOT, and Wi-Fi 6/Wi-Fi6E and has made way for smart laboratories, blood analyzers, and continuous glucose monitoring.
Our unique healthcare product development projects infused with the latest technological trends and strategies are ideal for your business.
BIOMARKERS
Biomarkers pave the path to digital healthcare product development
Different kinds of biomarkers help to identify diseases, but this blog focuses on those that can be traced through the electrical activity in the brain. These EEG biomarkers include:
Amplitude biomarkers: Thrown up by the intensity of EEG signals and indicating the brain frequency that prevails in a particular state.
Connectivity biomarkers: Uncovered through functional network connections between different regions in the brain that contribute to functions and disorders.
Entropy biomarkers: Shown by the complexity of the EEG signals and evaluating the health of the brain.
Event-related potentials: Revealed by time-locked EEG responses to specific events or stimuli, which provide an understanding of the cognitive processes.
Frequency biomarkers: Reflected in the distribution of brainwave frequencies in EEG data, including alpha, beta, theta, delta, and gamma.
Spectral power biomarkers: Quantified by the strength of the frequency bands and offering insights into brain activity and states -alert/sleepy.
Spikes and Epileptiform biomarkers: Detected by abnormal, sharp waveforms in EEG data and helping to identify epilepsy.
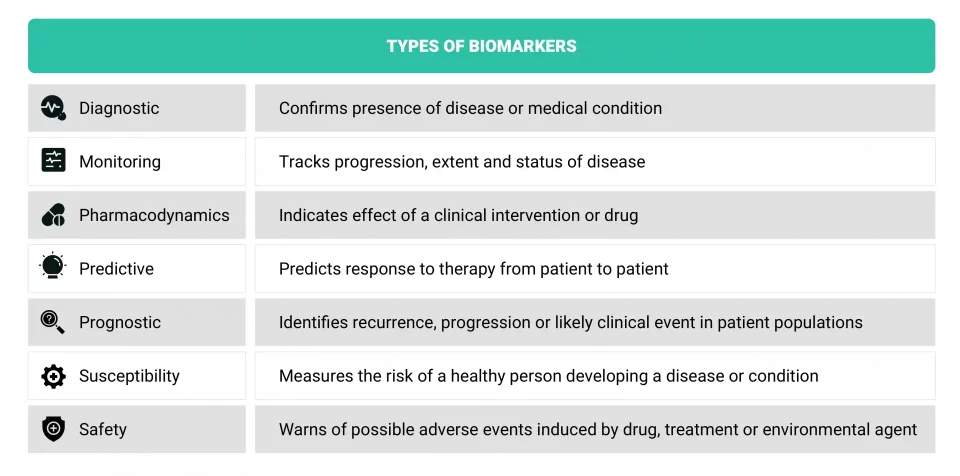 Different kinds of biomarkers can be deployed to develop digital healthcare technologies
Different kinds of biomarkers can be deployed to develop digital healthcare technologies
Beyond the known realms of brain activity, investigations are throwing up new biomarkers that healthcare software development companies leverage to innovate digital healthcare product development.
RESEARCH
Innovative uses of EEG biomarkers for digital healthcare product development
Let us look at the digital healthcare technologies (DHT) that employ new EEG biomarkers and their applications in new areas.
An EEG-based platform for titration of sedatives
One company is working on a pre-prototype of a software-as-medical-device (SaMD) with a graphical user interface. Based on a paper that linked specific EEG signals to postoperative delirium, it can interpret real-time EEG signals of a patient undergoing surgery. The EEG data is processed with a proprietary algorithm that can guide anesthetists on the choice of medication, the depth of anesthesia required, and the dosage. A patented feature is the ability of the DHT to decipher signals from older patients where cortical atrophy may have set in. Anesthetists will be able to assess and address the cognitive age of the brain, enabling patient-specific treatment based on brain health.
Depression drugs twinned with EEG biomarkers
A company is using an AI-powered biomarker platform to match drug development with specific patient groups to enable precision psychiatry. It has developed a pipeline of drugs that target post-traumatic stress disorder (PTSD) with the help of EEG activity, brain biomarkers, behavioral task performance, wearable data, and genetics. In a recent Phase 2 study, PSTD patients exhibiting a biomarker-defined cognitive profile demonstrated a greater improvement in CAPS-5 scores (that denote severity) than the patient group without the biomarker profile. This proves that while the diagnosis may be the same, the treatment has to be patient-specific; the validation of the biomarker across major depressive disorder populations also supports a transdiagnostic approach.
Managing pain with EEG biomarkers
Another company is working on a digital therapeutics platform that aims to modify pain sensitivity to manage chronic pelvic pain instead of opting for surgery or medications. The technology first analyzes a patient’s sensitivity to pain through EEG biomarkers and then reverses the triggers in the central nervous system. The company first built a proof of concept leveraging the biomarker to correct the mind-body connection for a variety of pain conditions. After the capsaicin-induced pain study, it was re-tested in a long-lasting elbow- pain study where the biomarker was assessed for stability across many patients. The validated platform for the neuro-modulation of pain sensitivity is being developed with a worldwide exclusive rights license.
Sleep monitoring EEG headband
An AI-assisted wearable headband uses integrated machine-learning algorithms to capture EEG data to monitor sleep architecture. Potential partners and clinical trial collaborators have been invited to participate in EEG-based sleep monitoring at home instead of a sleep laboratory or via wrist actigraphy. It is designed to record for a duration of 24 hours, is equipped with six electrodes, and features an integrated accelerometer to measure head movement and body position. Earlier, a clinical usability study demonstrated that it is well-tolerated by users and produces clinical-grade data in the home environment, facilitating decentralized clinical trials and remote monitoring.
We conduct extensive research to deploy our healthcare product development services.
CONSTRAINTS
The use of EEG datasets and digital biomarkers needs regulatory approvals
A biomarker is a defined characteristic measured as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to therapeutic interventions. Such data, when collected through digital patient engagement tools, are referred to as digital biomarkers when they explain, influence, or predict a health outcome.
Warning against the misuse of the term digital biomarker, the US Food and Drug Administration mandates a large body of evidence to “qualify” a biomarker for a single context of use. It has issued guidance and a framework under which it proposes to enhance review protocols as technologies develop. As the FDA is still building its own technical expertise in the digital healthcare product development arena, there are constraints.
- The FDA clearance criteria for software as a medical device and software in a medical device are still not clearly understood.
- The FDA has mandated that a software bill of materials be submitted along with a plan designed to “monitor, identify and address” possible cyber security issues of every cyber device and related medical system.
- Patient consent in sharing personal health information data may be one-time, whereas digital biomarkers evolve as more data is collected. Hence, updates to consent may be called for to get regulatory approval for a device.
- There is a fear that the bias of a human can seep into the machine learning algorithms built with skewed datasets.
- Digital healthcare product development with digital biomarkers may be undertaken when regulations are unclear, and so need to be future-proofed.
- Intellectual property concerns hinder transparency, and this may be a challenge for scrutiny and validation.
CONCLUSION
Digital biomarker-based healthcare technologies will evolve
The ball has started rolling, and digital biomarkers, in combination with EEG datasets, will transform predictive analytics in healthcare settings. As our approach to the management of disease and chronic health conditions evolves, the future promises early interventions, better patient engagement and health outcomes, and precision-based treatment protocols.
Our solutions are targeted to broaden your knowledge about patient data and outcomes.






